It is the electrons that are responsible for the chemical bavavior of atoms, and which identify the various chemical elements. In a neutral atom there are as many electrons as protons moving about nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. 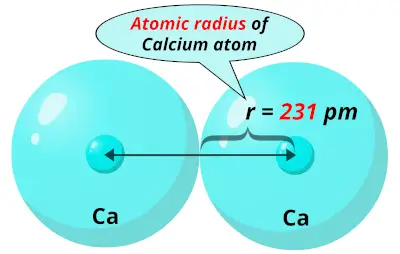
You can effortlessly find every single detail about the elements from this single Interactive Periodic table.The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Free Gift for you: Interactive Periodic Table Calcium is also used as a reducing agent in preparation of other different metals.Calcium is sometimes used as an alloying element with other metals to get the desired properties.Limestone is also used as a paint on walls.Limestone is also a calcium containing compound that is used in manufacturing cement for construction work.Calcium containing compounds (for example gypsum), is used in manufacturing various construction materials like POP (Plaster Of Paris).Calcium has many isotopes, out of which 40Ca is the most abundant isotope (and its abundance is around 97%).This is because the contact surface area of calcium increases with the water which makes the chemical reaction very fast. The powdered form of calcium reacts with water very rapidly.Calcium reacts with water and liberates hydrogen gas plus heat.Calcium is a chemically reactive metal so it is not found naturally, but it is always found as a compound with other elements.Chemical properties of CalciumĬhemical properties of calcium are mentioned below. Calcium salts can produce orange color, hence it is used in fireworks industries.Melting point of calcium is 842 ☌ and its boiling point is 1484 ☌.Calcium is a soft metal and it can be cut even with a kitchen knife.Calcium is a dull silvery grey colored soft alkaline earth metal.Physical properties of calcium are mentioned below. The physical and chemical properties of calcium element are mentioned below. Human body requires vitamin D to absorb the calcium from food.The requirement of calcium in the human body is fulfilled by eating (or drinking) calcium rich foods like yogurt, cheese, milk, spinach, etc.Calcium is an alkaline earth metal, so more calcium present in the soil results in alkaline soil (or soil having pH higher than 7). Calcium is also present in soil and it controls the pH level of soil.Out of all other metallic elements, the calcium element is the most abundant metallic element found in the human body.Calcium ions are also found dissolved in the sea water.Calcium is also the 5th most abundant element present in the human body.Calcium is the 5th most abundant element (by mass) found in the earth’s crust.Our teeth and bones contain calcium in it.Calcium is a very essential element for all living organisms.Interesting facts about calcium element are mentioned below. So the last electron of calcium enters the s-subshell or s-orbital. The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.įor example the electron configuration of calcium is 4s 2. How can you determine the blocks-wise position of elements? Protons in Calcium 20 Neutrons in Calcium 20 Electrons in Calcium 20 Symbol of Calcium Ca Atomic mass of CalciumĤ0.078 u Electrons arrangement in CalciumĢ, 8, 8, 2 Electronic configuration of Calcium 4s 2 Atomic radius of CalciumĢ31 picometers (van der Waals radius) Valence electrons in CalciumĢ 1st Ionization energy of Calcium 6.113 eV Electronegativity of Calciumġ.00 (Pauling scale) Crystal structure of CalciumįCC (Face centered cubic) Melting point of Calcium 1115 K or 842 ☌ or 1548 ☏ Boiling point of Calcium 1757 K or 1484 ☌ or 2703 ☏ Density of Calcium 1.55 g/cm 3 Main isotope of Calcium 40Ca Who discovered Calcium and when?īefore knowing this reason, first of all I want to ask you a simple question. Group: 2, Period: 4, Block: s Category of Calcium elementĪlkaline earth metals Atomic number of Calcium, or Let’s dive right into it! Calcium Element (Ca) Information Appearance of Calciumĭull silvery grey color State of Calcium at STP Solid Position of Calcium in Periodic table So if you want to know anything about Calcium element, then this guide is for you. In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Calcium element in Periodic table.) This is a SUPER easy guide on Calcium element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
